Immunofluorescence labeling with 2 primary antibodies on free-floating sections
CAT#
SI203
Category Immunohistostaining
Immunofluorescence labeling with 2 primary antibodies on free-floating sections
Description
This service includes tissue preparation, sectioning, immunostaining, mounting, coverslipping and labeling the slides. As a result, you will receive up to 60 immunostained sections per brain or per tissue block ready for microscopic observations.
Procedure: Following cryoprotection, tissue will be rapidly frozen in isopentane pre-cooled to -70°C. The frozen tissue will then be cut on a cryostat and collected in our unique section cryoprotection solution (cf. Products, Cat. #PC101). Subsequently, sections cut from various levels (or the levels of your option) will be processed free-floating for immunostaining with 2 specific antibodies according to the indirect immunofluorescence method¹ (cf. photo samples below).
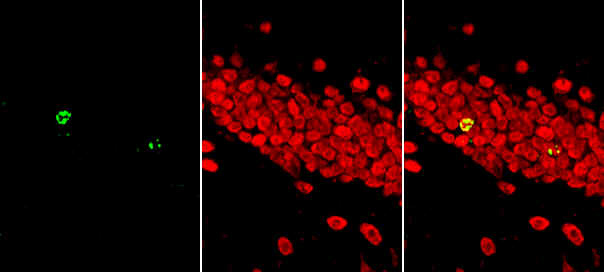 |
| Left: Cofocal image of BrdU-immunoreactivity. 30 µm cryostat section was cut from the hippocampal dentate gyrus of a mouse that survived for 24 hrs after the injection with 5-bromo-2-deoxyuridine (BrdU). This section was processed free-floating according to the indirect fluorescence method.
Middle: Cofocal image of NeuN-immunoreactivity. The same section as shown on the left was processed free-floating for NeuN-immunoreactivity according to the indirect fluorescence method. Note NeuN-labeled granule cells in the dentate gyrus. Right: Colocalization of BrdU- and NeuN-immunoreactivities. A digital overlay of the 2 images shown on the left. Note that the regions of colocalization, reflecting the additive effect of superimposed green and red pixels, appear in yellow. |
Remarks:
- A quotation is required before placing an order.
- The investigator needs to provide fixed tissue and the specific antibody.
- Please contact us for more information.
Reference:
- Coons, A.H. (1958) Fluorescent antibody methods. In J.F. Danielli (ed): General Cytochemical Methods. New York: Academic Press, pp. 399-422.
